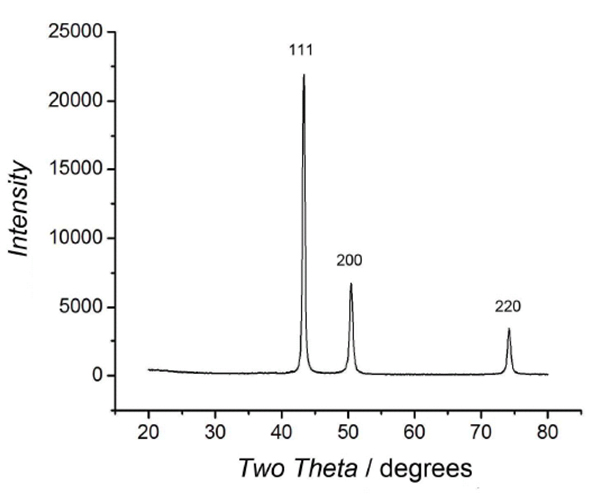
Therefore, because of multiple interactions between the polymer molecules and the metal surface, these polymer molecules cannot be easily detached from the surface. Polymers are unique candidates for protective layers of copper fine particles because polymer molecules attach to the particle surfaces at multiple points. The reported organic protective coating reagents, such as PVP, 23 CTAB, 28 oleic acids, 29 and alkane thiols, etc., 19 form a nonconductive shell around the surface. 27 For conductive inks and pastes, copper particles coated with organic polymers or molecules are highly useful because they can be readily dispersed in a suitable medium. One promising approach to prevent the oxidation of the particle surface is the application of a protective coating 19 of thin layers of polymers, 14,23–25 metal oxides, 26 or noble metals. To overcome the issue of copper nanoparticles and fine particle oxidation, many attempts have been made to improve the properties of the materials themselves and further develop appropriate sintering technologies. Additionally, in printed electronic materials, the presence of copper oxides on the particle surface is undesirable, increasing the required sintering temperature and reducing the electro-conductivity. 19–22 However, they can be oxidized, which limits their wide use. Therefore, in recent years, the synthesis of copper nanoparticles and fine particles has attracted substantial interest. Copper is an ideal material for industrial-scale printed electronics. In contrast, copper nanoparticles and fine particles are very attractive from the cost and anti-migratory perspectives. Conducting polymers may represent another possibility, but in many cases, their conductivity is not sufficient. The migration of silver is also an important issue that can degrade device performance. 17,18 However, high material costs limit their usage in large-scale printed electronic applications. Noble metals, especially silver nanoparticle inks, are highly stable and commonly used in conducting wires. Indeed, to produce flexible, lightweight, cost-effective and eco-friendly electronic devices, such as smart labels using radiofrequency identification (RFID), organic light-emitting diodes (OLEDs), and flexible-displays, printed electronic technologies will be frequently used. 12–15 Printing processes using these modern technologies are expected to facilitate the innovation of new manufacturing processes that will increase the productivity and reduce the required energy and resource consumption compared with the conventional processes. 12–16 In particular, conductive inks and pastes consisting of metal nanoparticles and fine particles are key materials in printed electronics because they can be used in low-cost wet printing processes. The steady increase in the use of nanoparticles and fine particles has resulted in their increased production and the development of handling and processing techniques for such materials.Īdditionally, the recent and remarkable progress in printed electronics is based on the development of useful materials, including nanomaterials. 1–4 Nanotechnology is currently used in diverse applications, including construction and composite materials, 5 catalysts, 6,7 antibacterial coatings, 8 biosensing applications, 9 plasmonic photo devices, 10 electrical devices, 11–13 and some consumer products. Introduction Owing to their unique properties, metal nanoparticles and fine particles have great potential to be used in developing new products. Our results strongly indicate that decantation under a nitrogen atmosphere can prevent the oxidation of copper fine particles but a slight oxidation was found. The surface oxidation state of the obtained copper fine particles could be considerably varied by altering the pH of the particle dispersion, as shown by X-ray diffraction and high-resolution transmission electron microscopy. After reduction, the fine particles were collected by decantation with pH control and washing, followed by drying at a moderate temperature. Metallic copper fine particles used for electro conductive pastes were prepared by the chemical reduction of cupric oxide microparticles in the presence of gelatin.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
